 |
 |
 |
 |
 |
 |
Chemical Probes for Epigenetics
Potent, selective and cell-permeable inhibitors of protein function ("chemical probes") are valued reagents in both fundamental and applied biological research, yet rarely available.
Chemical probes are essential for the early stages of drug discovery by allowing preclinical target validation in both academic and industrial laboratories: well-characterised inhibitors with defined mechanisms of action are important drivers of industrial drug discovery programmes.
Since 2008 the SGC has led an initiative to develop chemical probes that can selectively stimulate or block the activity of proteins involved in epigenetic control, complementing genetic knockouts and RNAi approaches to understand the cellular role of these proteins.
-
Continual generation of high-quality, open access chemical probes for epigenetics proteins (in excess of 14 probes, as of July 2013).
-
G9a methyltransferase (UNC0638 Nature Chemical Biology, 2011, UNC0642)
-
BET/ BRD4 (JQ1, Nature 2010)
-
BET (PFI-1, Cancer Research 2013 )
-
EZH2/1 (GSK343/UNC1999, ACS Med Chem Letters 2012 )
-
Lysine Demethylase/ JMJD3 & UTX (GSK-J1, Nature 2012)
-
Target Characterization
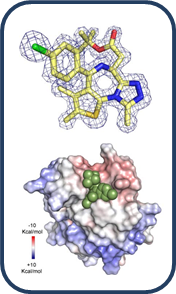 |
|
Structures
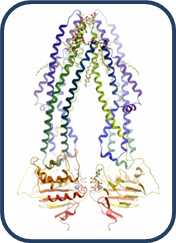 |
|
Recombinant Antibodies
- Helped coordinate Renewable Protein Binding Working Group (incorporating more than 11 institutions worldwide) that produced hundreds of antibodies targeting 20 SH2 domain proteins (Colwill et al, Nature Methods 2011).
- Launched a project to develop technologies, libraries and recombinant antibodies to human epigenetic proteins. The project is a carried out in partnership with several of the world leaders in the field (Sidhu, Kossiakoff, Dübel, Koide) and all output is to be made available without restriction on use.
- Generated single digit nM recombinant antibody and antibody-like reagents to over 100 human proteins.
