|
 |
 |
 |
 |
 |
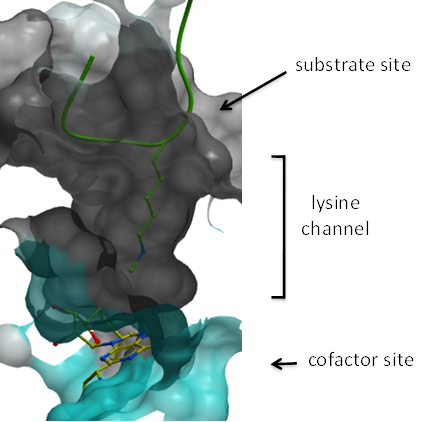
The PMT family is fairly large (64 members) and is composed of proteins that transfer a methyl group from the cofactor S-adenosylmethionine (SAM) to lysine and arginine side-chains of histone and non-histone substrates. Lysines can exist in three methylation states (mono, di, and tri-methylation) as can arginines (mono, sym-di, and asym-di-methylation). Most of the lysine methyltransferases contain a SET domain which is responsible for catalytic activity; an exception is DOT1L which is structurally closer to arginine methyltransferases. PMTs are quite specific about the sequence they methylate and the level of methylation is governed by the composition of the lysine-binding pocket. Inhibitors can be peptide-competitive, SAM-competitive, or allosteric (non-competitive with both SAM and peptide) and examples of all three inhibitor types have been co-crystallized with their targets.
Overexpression, gain-of-function mutation, translocation and recruitment at aberrant loci have been reported for many PMTs in diverse cancer types and inhibitors are being pursued as anti-cancer agents. Two classes of compounds have already reached the clinic: a DOT1L inhibitor and several EZH2 inhibitors.