|
 |
 |
 |
 |
 |
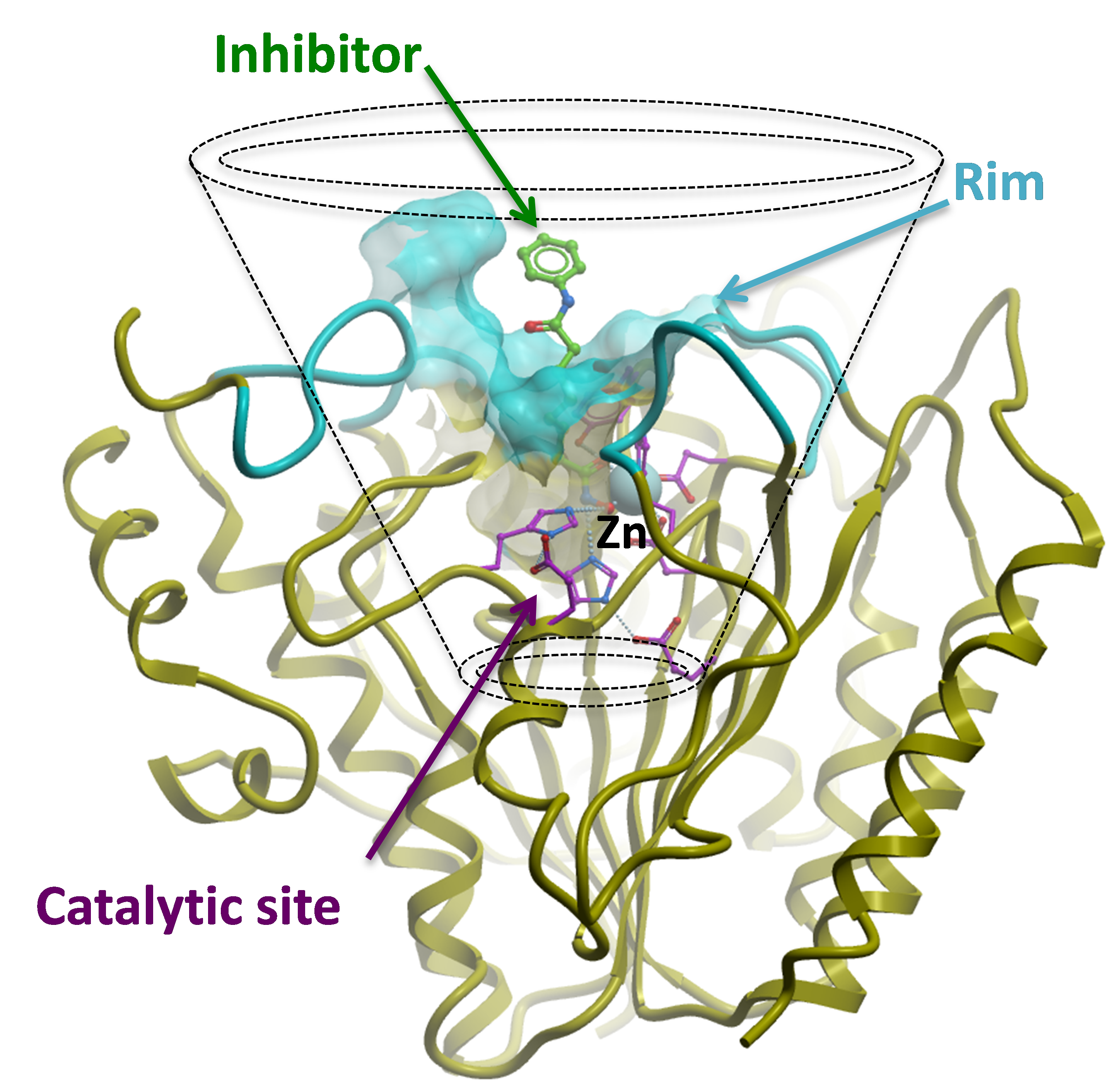
HDACs are divided into five phylogenetic classes. Enzymes from classes I, II and IV require a divalent metal ion for catalysis. Sirtuins (class III) are NAD+-dependent enzymes with protein deacetylase and ADP-ribosylase activity, and are structurally and biochemically unrelated to the other classes. HDACs deacetylate both histone and non-histone substrates. Several metal-dependent HDAC inhibitors are in the clinic; most of these target haematological malignancies. All HDAC inhibitors occupy the canonical acetyl-lysine channel of HDACs. A catalytic residue is absent in class IIa enzymes, which results in minimal deacetylase activity. Among sirtuins, deacetylase activity has been reported for SIRT1, SIRT2, SIRT3 and SIRT6. The most advanced compound is a SIRT1 inhibitor in clinical trials for Huntington’s disease.