|
 |
 |
 |
 |
 |
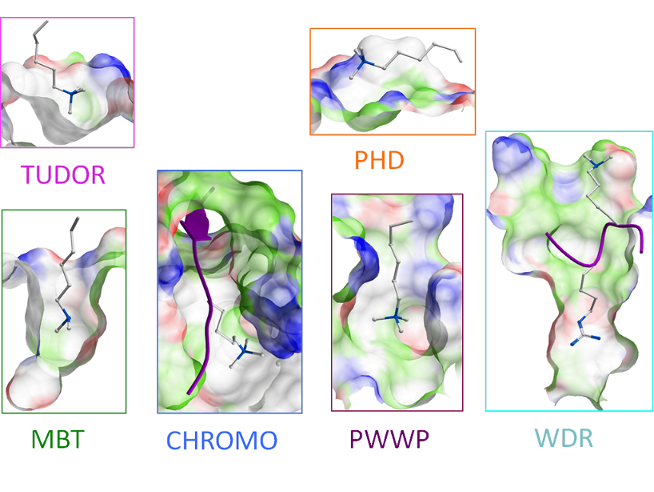
This is a large family of proteins which bind differently methylated lysine and arginine residues and can be divided into several sub-families.
Tudor Domains
Tudor domains commonly interact with di- and tri-methylated lysines and are present in proteins which typically contain additional active domains such as histone demethylases. These domains can occur twice in a protein sequence and are called tandem tudors.
MBT Domains
This is a fairly small family of 11 proteins which mostly bind mono- and di-methylated lysines. The MBTs are composed of multiple MBT-repeat sequences (2, 3, or 4) which only bind methyllysines when they act together. If the domains are expressed individually, no methyllysine binding occurs.
PHD Domains
This is a large family of proteins (104 members) which can bind H3 in various states of post-translational modification. H3K4me0/2/3 as well as H3R2me0/2 can bind to PHDs in addition to H3K14Ac. These proteins typically contain a zinc-binding motif which can also appear in duplicate (tandem PHD).
Chromo Domains
These are small domains of about 55 amino acids and are known to bind mono, di, and trimethyl H3K4/9/27.
PWWP
These proteins contain a MeLys binding pocket which is composed of four aromatic-containing amino acids as the name implies. All PWWP domains commonly occur in proteins containing other motifs such as HMT and PHD.
WD
The WD protein family is very large, however, only 18 of these are known to be involved in chromatin binding. These proteins contain a single or multiple propeller-type structures with 7 “vanes”.