Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: MAT2BB-c018 |
GenBank GI number: gi|33519455 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Amplified construct sequence:
CTTAAGAAGGAGATATACTATGAACC
GTCGCGTTCTGGTTACCGGTGCTACT
GGTCTGCTGGGTCGTGCAGTACACAA
GGAGTTTCAGCAGAATAACTGGCACG
CTGTAGGTTGCGGTTTTCGTCGTGCG
CGTCCAAAATTCGAGCAAGTAAACCT
GCTGGACTCTAACGCTGTTCACCACA
TCATCCACGATTTCCAGCCGCACGTT
ATTGTGCATTGCGCTGCTGAACGTCG
TCCTGACGTTGTAGAAAACCAGCCAG
ATGCTGCCTCCCAACTGAACGTTGAT
GCGTCTGGTAACCTGGCCAAGGAAGC
TGCTGCAGTAGGCGCATTTCTGATCT
ATATCAGCTCCGATTACGTTTTCGAC
GGTACCAACCCGCCTTATCGTGAGGA
AGACATCCCGGCTCCACTGAACCTGT
ACGGTAAGACGAAGCTGGATGGCGAA
AAAGCGGTGCTGGAAAATAACCTGGG
TGCTGCAGTTCTGCGTATCCCGATCC
TGTATGGTGAAGTCGAAAAACTGGAA
GAATCCGCGGTTACTGTTATGTTCGA
CAAGGTCCAGTTCTCCAATAAGTCTG
CAAACATGGACCATTGGCAACAGCGC
TTCCCGACTCACGTTAAAGATGTGGC
TACGGTTTGCCGCCAACTGGCAGAGA
AGCGTATGCTGGACCCGAGCATCAAG
GGTACTTTCCATTGGAGCGGCAACGA
ACAGATGACGAAATACGAAATGGCAT
GTGCGATTGCCGACGCATTCAACCTG
CCATCTTCTCACCTGCGTCCGATCAC
CGATTCCCCAGTGCTGGGTGCACAAC
GTCCACGTAATGCGCAACTGGACTGT
TCTAAACTGGAAACTCTGGGTATCGG
TCAGCGCACTCCATTTCGTATCGGTA
TTAAGGAGTCTCTGTGGCCGTTCCTG
ATCGATAAACGTTGGCGTCAGACTGT
GTTCCATGCAGAGAACCTCTACTTCC
AATCGCACCATCATCACCACCATGAT
TACAAGGATGACGACGATAAGTGAGG
ATCC
|
Final protein sequence (Tag sequence in lowercase):
MNRRVLVTGATGLLGRAVHKEFQQNN
WHAVGCGFRRARPKFEQVNLLDSNAV
HHIIHDFQPHVIVHCAAERRPDVVEN
QPDAASQLNVDASGNLAKEAAAVGAF
LIYISSDYVFDGTNPPYREEDIPAPL
NLYGKTKLDGEKAVLENNLGAAVLRI
PILYGEVEKLEESAVTVMFDKVQFSN
KSANMDHWQQRFPTHVKDVATVCRQL
AEKRMLDPSIKGTFHWSGNEQMTKYE
MACAIADAFNLPSSHLRPITDSPVLG
AQRPRNAQLDCSKLETLGIGQRTPFR
IGIKESLWPFLIDKRWRQTVFHaenl
yfqshhhhhhdykddddk |
Tags and additions: C-terminal His-tag with TEV protease cleavage site |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: 10µl of a glycerol stock was inoculated into 5ml of TB medium (supplemented with 50µg/ml Kanamycin, 34µg/ml Chloramphenicol) and cultured at 37°C o/n in a shaking incubator (275rpm). Next day 0.75 ml of o/n culture was used to inoculate 1 litre of TB medium (6x) and grown at 37°C with vigorous shaking (160 rpm) until the culture reaches an OD600 of 1.4. Temperature was reduced to 18°C, and cells were induced with IPTG at a concentration of 0.5 mM, and further cultivated for 16 hrs. Cells were harvested by centrifugation at 6500rpm for 10 min, and the cell pellet was stored at -20°C until further use.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5 mM Imidazole; 5% Glycerol; Complete® protease inhibitors (Roche, 1 tbl/50ml).
Extraction buffer, extraction method: Frozen pellets were thawed and resuspended in a total volume of 30-40ml of lysis buffer, and disrupted by using Avestin C-5 microfluidizer, and a supernatant containing the target protein was obtained by centrifugation at 21,000rpm for 45 minutes. |
Column 1: Ni-Sepharose 6 Fast Flow |
Column 1 Buffers:
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP; 5 mM Imidazole.
Wash buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP; 30 mM Imidazole.
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP; 250 mM Imidazole. |
Column 1 Procedure: The column was packed with 2ml of Ni-Sepharose 6 Fast Flow slurry and equilibrated with 15ml of binding buffer. The supernatant was loaded onto the column and the column was washed with 20ml of binding buffer and then 20ml of washing buffer. The protein was eluted with 10ml of elution buffer. |
Column 2: Superdex S200 16/60 HiLoad (GE/Amersham) |
Column 2 Buffer: 10 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP. |
Column 2 Procedure: The eluted protein from the Ni-affinity column was loaded on the gel filtration column in GF buffer at 1.0ml/min on an ÄKTA Purifier system. Eluted protein was collected in 1ml fractions. |
Enzymatic treatment: TEV cleaved. |
Column 3: Ni-Sepharose (TEV Clean up) |
Column 3 Buffer: 10 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP. |
Column 3 Procedure: Total 10mg of protein was cleaved with 600µg of TEV protease at 4°C for 48 hours. |
Mass spectrometry characterization: Corresponds to theoretical mass, as determined by ESI-TOF MS. |
Protein concentration: Protein was concentrated to 10mg/ml using an Amicon 3kDa cut-off concentrator. |
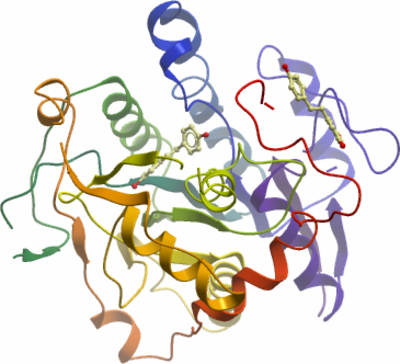
Crystallisation: Crystals were grown by vapour diffusion in sitting drops at 4°C. Before setting up the experiment 5 mM NADP and 2 mM resveratrol were added to the protein. A sitting drop consisting of 100nl protein and 200nl well solution was equilibrated against well solution containing 1.05 M Lithium sulphate and 0.45 M TMAO (Trimethylamine N-oxide). The crystals were mounted directly from the drop using 25% ethylene glycol as a cryoprotectant and flash-cooled in liquid nitrogen. |
Data collection: Resolution: 2.80Å. Phasing: Synchrotron SLS-X10SA, single wavelength. |