Molecular Biology
Entry Clone Accession: IMAGE:3345801
Entry Clone Source: MGC
SGC Construct ID: MAGEA3A-c203
Protein Region: E104-E314
Vector: pNH-TrxT. This vector adds his6 and thioredoxin sequences, followed by a TEV protease cleavage site, at the N-terminal of the protein.
Host: BL21(DE3)-R3-pRARE2
Sequence (with tag(s)): MHHHHHHSSGMSDKIIHLTDDSFDTDVLKADGAILVDFWAEWCGPCKMIAPILDEIADEYQGKLTVAKLNIDQNPGTAPKYGIRGIPTLLLFKNGEVAATKVGALSKGQLKEFLDANLAGTENLYF*QSMEFQAALSRKVAELVHFLLLKYRAREPVTKAEMLGSVVGNWQYFFPVIFSKASSSLQLVFGIELMEVDPIGHLYIFATCLGLSYDGLLGDNQIMPKAGLLIIVLAIIAREGDCAPEEKIWEELSVLEVFEGREDSILGDPKKLLTQHFVQENYLEYRQVPGSDPACYEFLWGPRALVETSYVKVLHHMVKISGGPHISYPPLHEWVLREGEE
Sequence after tag cleavage: SMEFQAALSRKVAELVHFLLLKYRAREPVTKAEMLGSVVGNWQYFFPVIFSKASSSLQLVFGIELMEVDPIGHLYIFATCLGLSYDGLLGDNQIMPKAGLLIIVLAIIAREGDCAPEEKIWEELSVLEVFEGREDSILGDPKKLLTQHFVQENYLEYRQVPGSDPACYEFLWGPRALVETSYVKVLHHMVKISGGPHISYPPLHEWVLREGEE
DNA Sequence: CATATGCACCATCATCATCATCATTCTTCTGGTATGAGCGATAAAATTATTCACCTGACTGACGACAGTTTTGACACGGATGTACTCAAAGCGGACGGGGCGATCCTCGTCGATTTCTGGGCAGAGTGGTGCGGTCCGTGCAAAATGATCGCCCCGATTCTGGATGAAATCGCTGACGAATATCAGGGCAAACTGACCGTTGCAAAACTGAACATCGATCAAAACCCTGGCACTGCGCCGAAATATGGCATCCGTGGTATCCCGACTCTGCTGCTGTTCAAAAACGGTGAAGTGGCGGCAACCAAAGTGGGCGCACTGTCTAAAGGTCAGTTGAAAGAGTTCCTCGACGCTAACCTGGCCGGTACCGAGAACTTGTACTTCCAATCCATGGAGTTCCAAGCAGCACTCAGTAGGAAGGTGGCCGAGTTGGTTCATTTTCTGCTCCTCAAGTATCGAGCCAGGGAGCCGGTCACAAAGGCAGAAATGCTGGGGAGTGTCGTCGGAAATTGGCAGTATTTCTTTCCTGTGATCTTCAGCAAAGCTTCCAGTTCCTTGCAGCTGGTCTTTGGCATCGAGCTGATGGAAGTGGACCCCATCGGCCACTTGTACATCTTTGCCACCTGCCTGGGCCTCTCCTACGATGGCCTGCTGGGTGACAATCAGATCATGCCCAAGGCAGGCCTCCTGATAATCGTCCTGGCCATAATCGCAAGAGAGGGCGACTGTGCCCCTGAGGAGAAAATCTGGGAGGAGCTGAGTGTGTTAGAGGTGTTTGAGGGGAGGGAAGACAGTATCTTGGGGGATCCCAAGAAGCTGCTCACCCAACATTTCGTGCAGGAAAACTACCTGGAGTACCGGCAGGTCCCCGGCAGTGATCCTGCATGTTATGAATTCCTGTGGGGTCCAAGGGCCCTCGTTGAAACCAGCTATGTGAAAGTCCTGCACCATATGGTAAAGATCAGTGGAGGACCTCACATTTCCTACCCACCCCTGCATGAGTGGGTTTTGAGAGAGGGGGAAGAGTGACAGTAAAGGTGGATACGGATCCGAATTCGAGCTCCGTCGACAAGCTT
Protein Expression
Medium: Terrific Broth
Antibiotics: Kanamycin, 50 µg/ml
Procedure: The expression plasmid was transformed into the host strain and plated on LB-agar containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. Several colonies were combined to inoculate a 1-ml culture in TB (+ 50 µg/ml kanamycin, 35 µg/ml chloramphenicol). The culture was grown overnight, glycerol was added to 15% v/v (from a 60% stock), and the resulting glycerol stock was frozen at -80°C. A loopful of cells from the glycerol stock was inoculated into 100-ml of TB medium containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol and grown overnight at 37°C.
6 x 1 L of TB medium (+ 50 µg/ml kanamycin) in 2.5L UltraYield baffled flasks were inoculated with 10 ml/L of the overnight culture. The cultures were grown at 37°C until OD600 of 0.2 and then shifted to 18°C. After 30 minutes, IPTG was added to 0.3 mM, and growth continued overnight. The cells were collected by centrifugation, the pellets were scraped out and transferred to 50-ml Falcon tubes and frozen at -80°C.
Protein Purification
Buffers:
Lysis buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol,10 mM imidazole, 0.5 mM TCEP,
Affinity buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 10 mM imidazole, 5% glycerol, 0.5 mM TCEP;
Wash buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 45 mM imidazole, 5% glycerol, 0.5 mM TCEP;
Elution buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 300 mM imidazole, 5% glycerol, 0.5 mM TCEP.
GF buffer: 10 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP.
Procedure:
NOTE: This MAGEA3 protein construct migrates faster on SDS-PAGE than expected, as a band of ~18 kDa. The correct mass of the protein has been confirmed as 24 kDa by mass spectrometry.
Frozen cell pellets were thawed briefly in a bath of warm water (20 - 37°C) then transferred to ice. The cells were suspended in 100 ml of lysis buffer and disrupted by sonication. Nucleic acids and cell debris were removed by centrifugation for 20 minutes at 25,000 xg.
Column 1: The cell extract was applied to 3 ml Ni-IDA sepharose in a gravity flow column. The column was then washed with 10 volumes of affinity buffer, 10 column volumes (CV) of wash buffer, then eluted with 5 CV of elution buffer.
Tag removal: TEV protease (1 mg / 50 mg protein by Bradford assay) was added to the protein, which was placed in a dialysis tube (3.5 kDa cutoff, Snakeskin membrane) and dialyzed overnight at 4°C against GF buffer. The His8-tagged TEV protease and contaminating proteins were removed by reapplication of dialysed proteins to a Ni-IDA IMAC column (2 ml CV).
Column 2: Proteins passing through the column were pooled and concentrated, using a 10 kDa MWCO centrifugal concentrator, to 1ml before loading onto a HiLoad 16/60 Superdex S75 gel filtration column equilibrated in GF buffer.
Column 3: Fractions containing MAGE-A3 were pooled and diluted 10 fold in H2O to 50 mM NaCl and applied to a 1ml Mono Q anion exchange column equilibrated in 50 mM Hepes pH 7.5, 50mMNaCl. Proteins were eluted with a linear gradient to 50 mM Hepes pH7.5, 1M NaCl over 30 CV, and fractions containing MAGE-A3 were pooled and concentrated. The protein was confirmed by ESI-TOP intact mass spectrometry (Predicted: 24094.9; observed: 24094.9)
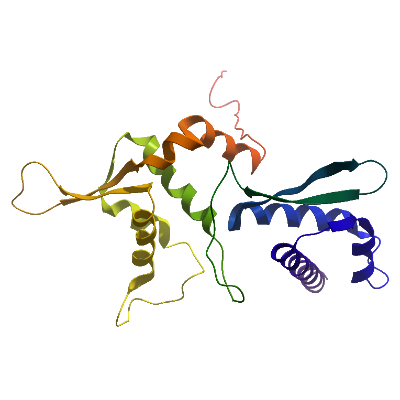
Structure Determination
Crystallization: For crystallization, MAGE-A3 was concentrated to 5 mg/ml and sitting drop vapour diffusion crystallization trials were set up with a Mosquito (TTP Labtech) crystallisation robot. MAGE-A3 crystallized at 4°C in conditions containing 15% PEG 3350, 0.1M Mg-Formate. Crystals were loop mounted and-cryo protected in a reservoir solution supplemented with 25% ethylene glycol
Data Collection: Data were collected to 2.0Å resolution at Diamond light source beamline I24, and processed using XDS
Data Processing: The structure was solved by molecular replacement using the program PHASER and the structure of MAGE-A4 as a search model. Refinement was performed using PHENIX REFINE to a final Rfactor = 22.3%, Rfree = 23.4%.