Expressed almost exclusively in proliferating cells such as thymus, liver, and bone marrow, the transcription factor ICBP90 (UHRF1) regulates expression of checkpoint related proteins and tumor suppressors. It contains a ubiquitin-like (UbL), PHD zinc finger, SRA and RING finger domain. The SRA domain has been shown to bind symmetric methylated-CpG dinucleotides sites, including the inverted CCAAT regions of the topoisomerase II alpha gene. Other ICBP90 promoter binding sites include those of retinoblastoma, p16ink4a, and p14arf. The SRA domain also functions to recruit HDAC1, which depresses transcription through chromatin remodeling. On the other hand, the ring finger recruits loaded E2’s for ubiquitylation of histones, which has the effect of decreasing local histone concentration thereby increasing transcriptional activity. Moreover, phosphorylation of ICBP09 by CK2 is thought to contribute to the normal mechanism of cell cycle progression and well as normal mechanism of proliferation.
Consistent with its tissue distribution in normal cells, ICBP90 is overexpressed in carcinomas, including breast cancer, pancreatic adenocarcinoma, cervical cancer, and numerous tumorigenic cell lines. Although normally downregulated by p53 dependant DNA damage, ICBP90 concentrations are high in cancer cells, where transcription of pRB is low and the concentration of the pRB-EF2-1 complex is low (the latter inhibits cell cycle progression). Concomitantly, tumor suppressor proteins are also downregulated by ICBP90 associated HDAC1. In T Cell models, EF2-1, a transcriptional activator of ICBP90, plays a prominent role. TCR mediated regulation of EF2-1 leads to higher transcription of pRB, which inhibits cell cycle progression and promotes apoptosis.
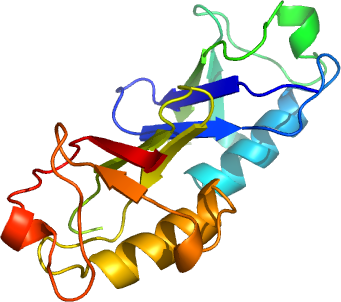
The amino terminal ubiquitin like domain is 35% identical with human ubiquitin and unlikely to be transferable. Instead, it may act as a targeting, protein/protein interaction domain. To begin functional characterization, we have determined the high resolution structure of the ubiquitin-like domain of human ICBP90. It has the typical alpha/beta ubiquitin fold, with an RMS deviation of 0.907 Angstroms when comparing 73 alpha carbons of human ubiquitin (1UBI). Of three surface lysines (K26, K33, and K52), K33 and K52 are structurally conserved with those of ubiquitin (K29 and K48); polyubiquitylation of the latter being the key signal for proteasomal protein turnover. The surface of ICBP90 corresponding to the protein/protein binding hotspot of ubiquitin (loosely centered on I44 of ubiquitin) is similarly composed of a hydrophobic patch (F48, L74, and M10) surrounded by charged or polar residues.