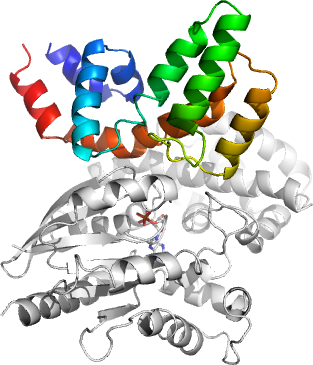
XX02RGS8A: Human regulator of G-protein signalling 8 in complex with Galphai3
PDB:2ODE
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:RGS8A-s001
Entry Clone Source:MGC
SGC Clone Accession:Tag:N-terminal hexahistidine tag
Host:BL-21(DE3)R3 (phage resistant)
Construct
Prelude:Sequence:SMLSTEEATRWADSFDVLLSHKYGVAAFR AFLKTEFSEENLEFWLACEEFKKTRSTAK LVSKAHRIFEEFVDVQAPREVNIDFQTRE ATRKNLQEPSLTCFDQAQGKVHSLMEKDS YPRFLRSKMYLDLLSQSQRRLS
Vector:pLIC- SGC1
Growth
Medium:Antibiotics:Procedure:Purification
ProcedureColumn 1: Low pressure chromatography using Bio-Rad Econo column (2.5 cm x 13 cm).
Procedure: Total volume of Ni-NTA added to BioRad drip column: 4 mls (50 %). Resin washed with 12.5 ml of WB1. The supernatent was applied to a column using 5 ml pipette and allowed to pass over the resin. The flow through was collected in a 50 ml falcon tube and applied once more to the column. Two wash steps followed. Wash with 12.5 ml of WBI. Wash with 12.5 ml column vols of WBII. Elute with 14 mls of EB into 7x2 ml fractions.
Column 2 : Size exclusion using a S200 16/60 column
Procedure: The column was pre-equilibrated with two column volumes of GF buffer (flow rate 1 ml/min). The fractions from gel filtration that contained RGS8A were pooled and concentrated before loading on to S200 16/60 gel filtration column. The fractions containing protein were identified on a coomasie blue stained gel.
Enzymatic treatment : His-Tag removal. The eluate was treated with Tev protease. The sample was rebound to Ni sepharose to remove Tev and other contaminants. The His tag cleaved protein was concentrated to 300 µM.
Extraction
Procedure1 tablet protein inhibitor in 10 ml Lysis Buffer was added to the 1L growth pellet. Total vol: 45 mls (estimate). Cell breakage: 3 passes through the Emulsiflex C5 high pressure homogeniser. Centrifuge for 45 mins at 16000 rpm and 4°C to remove cell debris. Discard pellet.
Concentration:LigandMassSpec:The expected mass of RGS8A-C007 without histidine tag is 16195.2. The experimentally determined mass was 16195.17.
Crystallization:NMR Spectroscopy:Data Collection:Data Processing: