The probe GSK-LSD-1 (hydrochloride) is available from Cayman Chemical, Sigma and Tocris.
| Probe |
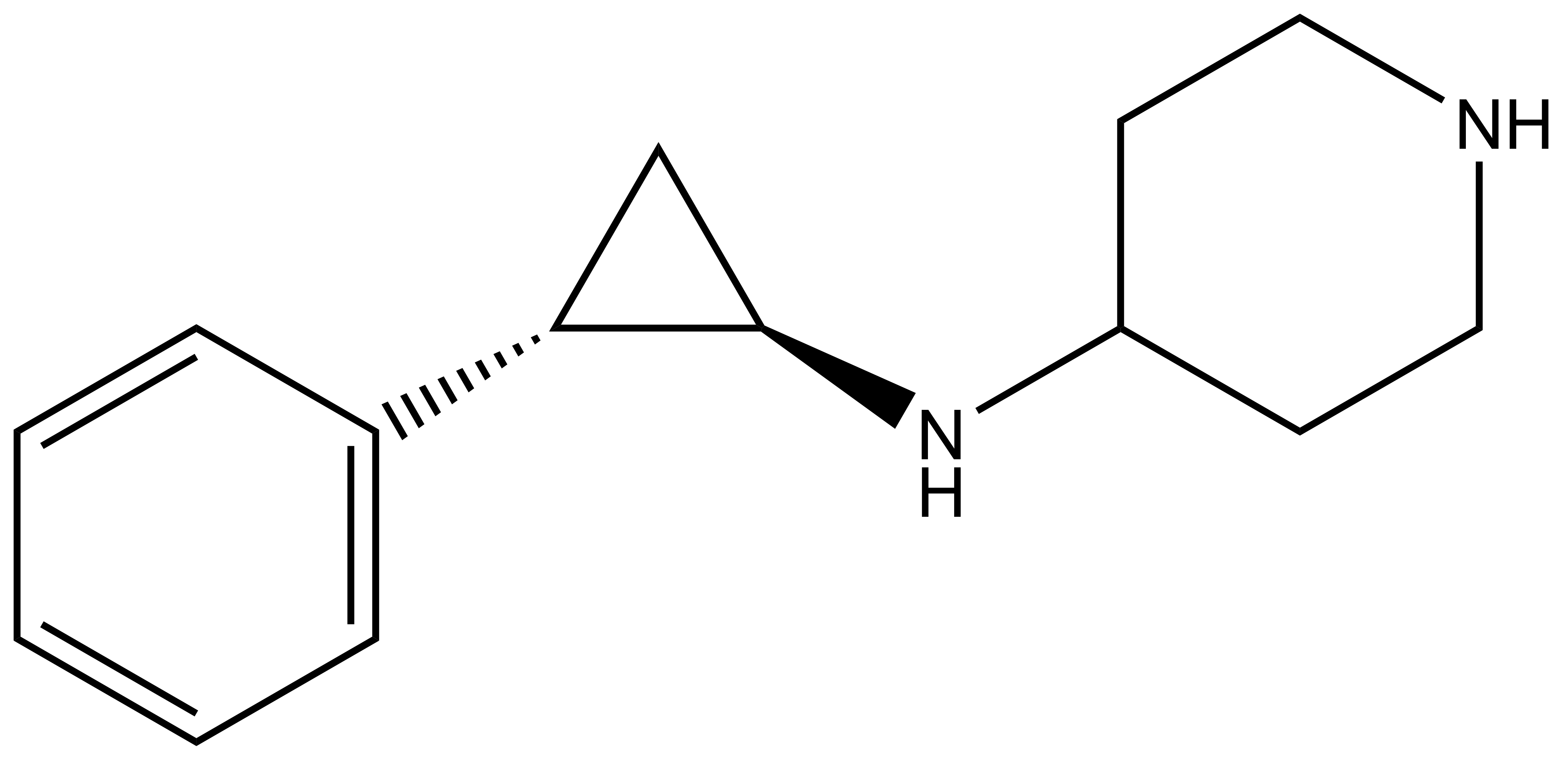 |
GSK-LSD1 |
GSK-LSD1 (trans-racemic)
MW 216.3
FW 289.2 (di-HCl)
Lysine specific demethylase 1 (LSD1) is a histone demethylase found in various transcriptional co-repressor complexes. LSD1 is involved in ES cell differentiation, hematopoiesis, and has been described as having a role in Acute Myeloid Leukemia (AML). GlaxoSmithKline has developed an irreversible, mechanism based inhibitor of LSD1, GSK-LSD1, and has made this available as a chemical probe as part of the SGC epigenetics initiative. GSK-LSD1 inhibits LSD1 with an IC50 of 16 nM and is > 1000 fold selective over other closely related FAD utilizing enzymes (i.e. LSD2, MAO-A, MAO-B). GSK-LSD1 induces gene expression changes in cancer cell lines (average EC50 < 5 nM) and inhibits cancer cell line growth (average EC50 < 5 nM).
LSD-1 at 10 µM showed no activity against 55 human recombinant receptors in the CEREP ExpresSProfile selectivity/specificity profile (GPCR, transporters, ion channels), except 5-HT1A (49% inhibition) 5-HT transporter (74% inhibition) and dopamine transporter (39 % inhibition).
| Probe |
 |
GSK-LSD1 |
| Physical and chemical properties for GSK-LSD1 | |
| Molecular weight | 216.2 |
| Molecular formula | C14H20N2 |
| IUPAC name | N-(2-phenyl-cyclopropyl)-piperidin-4-amine |
| MollogP | 2.639 |
| PSA | 23.66 |
| No. of chiral centres | 2 |
| No. of rotatable bonds | 3 |
| No. of hydrogen bond acceptors | 2 |
| No. of hydrogen bond donors | 2 |