GSK6853 is available for purchase from Cayman Chemical, Sigma and Tocris.
Its negative control (GSK9311) is available for purchase from Sigma and Tocris (hydrochloride).
| Probe | Negative control | |
 |
|  |
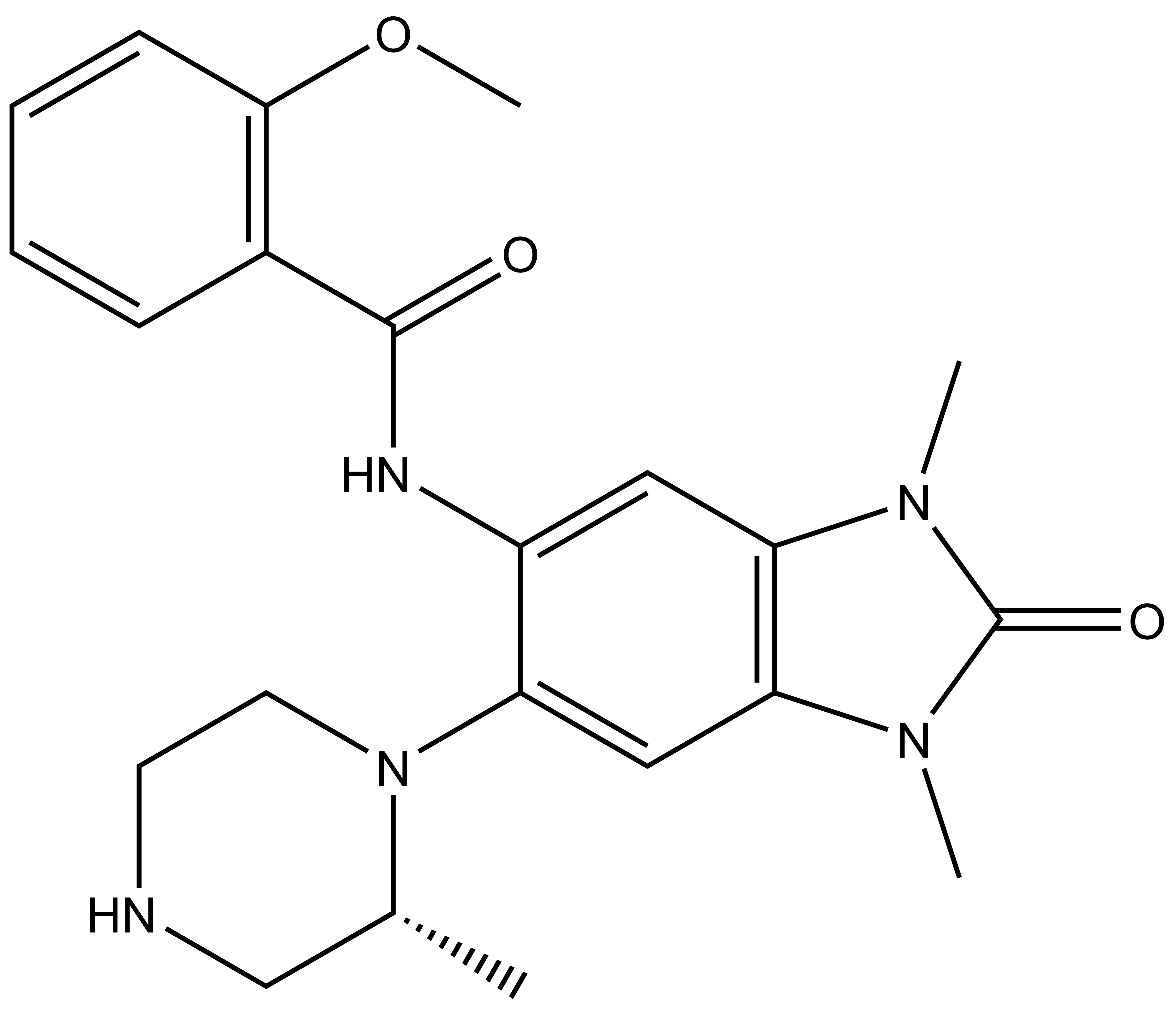
GSK6853 |
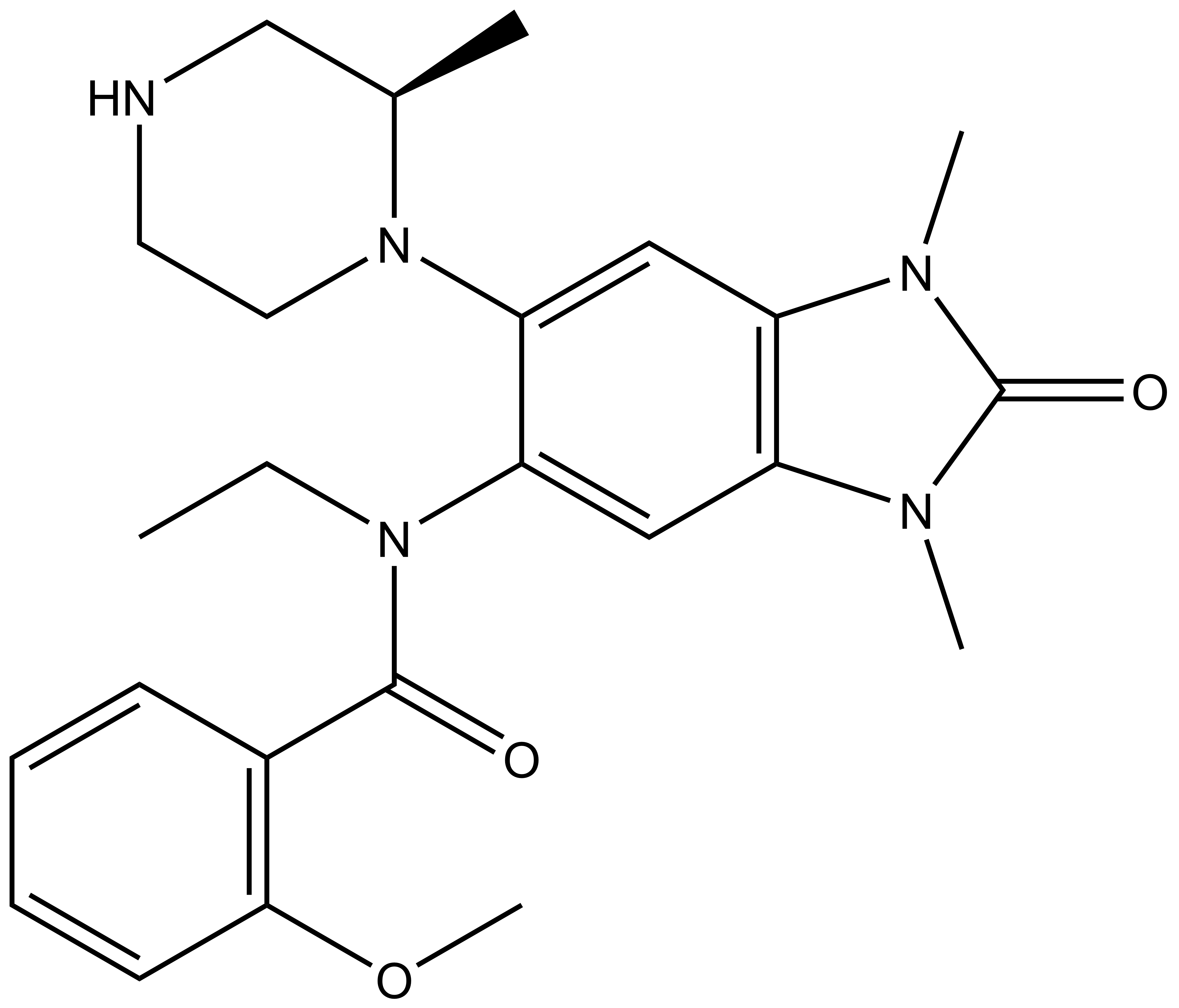
| GSK9311 |
A chemical probe for the bromodomains of the BRPF (BRomodomain and PHD Finger containing) family of proteins (BRPF1/2/3) has been discovered by the SGC.
BRPF1, BRPF2 (BRD1) and BRPF3 are scaffolding proteins, assembling HAT complexes of the MOZ/MORF family (MOZ, Ybf2/Sas3, Sas2, and Tip60) (1). These MYST complexes have a tetrameric core containing BRPF, the tumour suppressor ING and Eaf6/EPC (enhancer of polycomb)-related scaffold subunits. MYST complexes play crucial roles in DNA repair, recombination, and replication as well as in transcription activation (2,3). MOZ is frequently translocated in AML (acute myeloid leukemia) and is required for HSC proliferation (4).
Two BRPF1 isoforms (isoform A and B) can be generated by alternative splicing. In contrast to BRPF1B, the isoform A harbours a residue insertion in the ZA-loop that prevents binding to acetylated histone peptides (5).
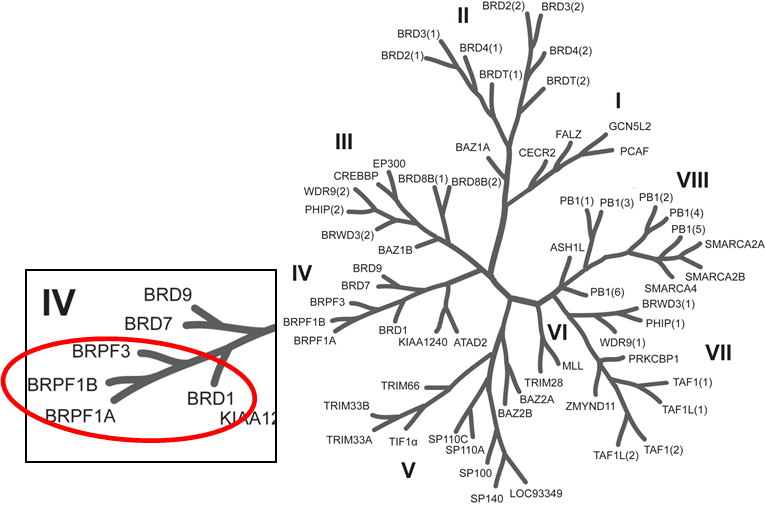
Phylogenetic tree of bromodomains and detailed view at the BRPF family.
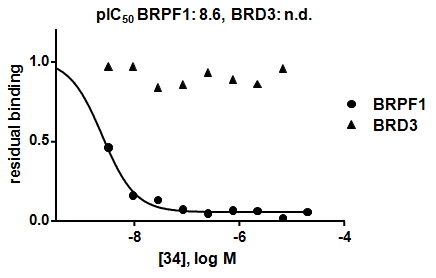
GSK6853 has excellent BRPF1 potency with an IC50 of 8 nM in a TR-FRET assay and a KD of 0.3 nM as determined by Bromoscan. In a panel of 48 bromodomains GSK6853 shows a greater than 1600-fold selectivity over all other bromodomains. In addition, GSK6853 exhibits potent binding to full-length endogenous BRPF1 (pIC50 of 8.6 nM) in a chemoproteomic competition binding assay.
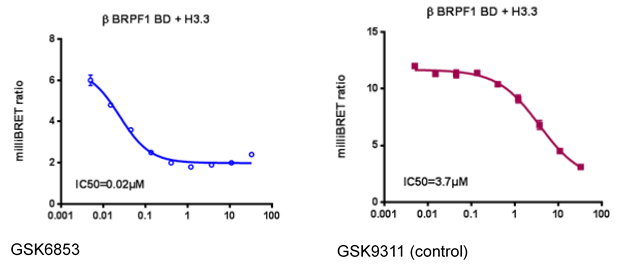
A NanoBRETTM cellular target engagement assay evaluating the interaction of BRPF1B with histones suggested IC50 of 20 nM for GSK6853.
To minimize the chance of off-target effects we recommend that a concentration of no higher than 1 μM should be used in cell-based assays.
In a NanoBRETTM cellular target engagement assay using isolated BRPF1B BRD with NLS and Halo-tagged histone H3.3 BRPF1 showed a dose-dependent displacement from histone H3.3, with cellular IC50 of 20 nM.
Chemoproteomic competition binding assay in HUT-78 cell lysate shows binding to endogenous BRPF-1 with selectivity over BRD3.
| Chrom logD pH 7.4 | 2.0 |
| CLND solubility (μg/mL) | 140 |
| iv CLb (mL/min/kg)/t1/2 (h) | 107/1.7 |
| F% ip/po (3 mg/kg) | 85/22 |
| Probe | Negative control | |
 |
|  |
GSK6853 |
| GSK9311 |
| Physical and chemical properties for GSK-6853 | |
| Molecular weight | 409.2 |
| Molecular formula | C22H27N5O3 |
| IUPAC name | 3-((2-methoxy-phenyl)-formylamino)-7,9-dimethyl-4-(2-methyl-piperazin-1-yl)-7,9-diaza-bicyclo[4.3.0]nona-1,3,5-trien-8-one |
| MollogP | 1.779 |
| PSA | 62.28 |
| No. of chiral centres | 1 |
| No. of rotatable bonds | 5 |
| No. of hydrogen bond acceptors | 6 |
| No. of hydrogen bond donors | 2 |
| Physical and chemical properties for GSK-9311 (Negative Control) | |
| Molecular weight | 437.2 |
| Molecular formula | C24H31N5O3 |
| IUPAC name | 3-(ethyl-((2-methoxy-phenyl)-formyl)-amino)-7,9-dimethyl-4-(2-methyl-piperazin-1-yl)-7,9-diaza-bicyclo[4.3.0]nona-1,3,5-trien-8-one |
| MollogP | 2.369 |
| PSA | 54.93 |
| No. of chiral centres | 1 |
| No. of rotatable bonds | 6 |
| No. of hydrogen bond acceptors | 6 |
| No. of hydrogen bond donors | 1 |
Temperature Shift
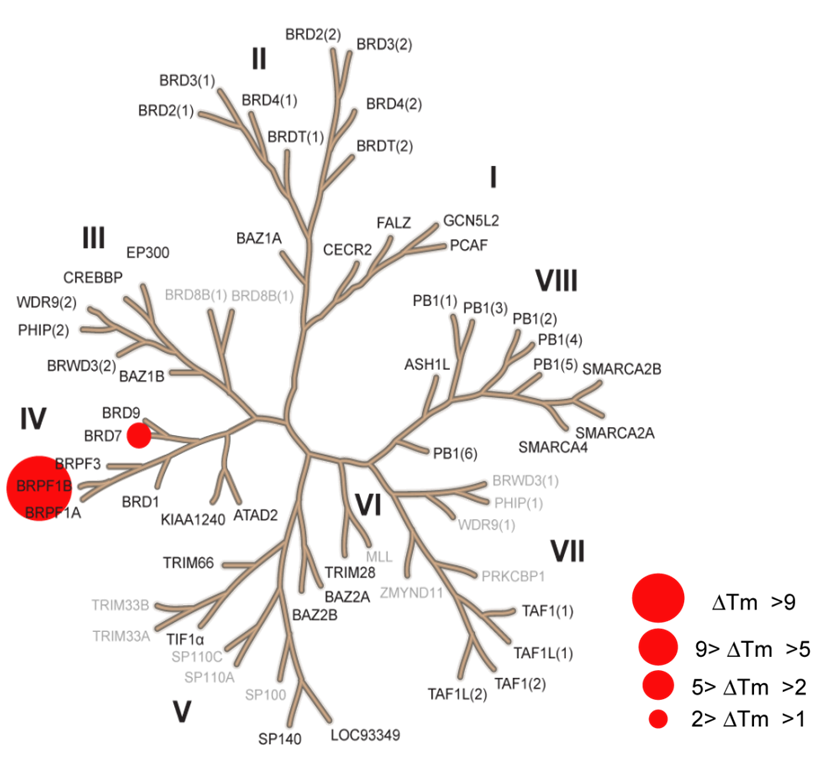
The temperature shifts mapped onto the phylogenetic tree using red circles corresponding to ΔTm as indicated in the figure.
BROMO scanTM

Bromodomain selectivity was evaluated in the BROMO scan™ panel (DiscoveRx Corp., Fremont, CA, USA, http://www.discoverx.com). This screen measures competition against reference immobilized ligands for 35 DNA-tagged bromodomains. GSK6853 is more than 1600 selective for BRPF1B over tested bromodomains.
GSK6853 was tested in a panel of 48 unrelated targets only weak off-target activity has been observed compared to the activity on BRPF1B.
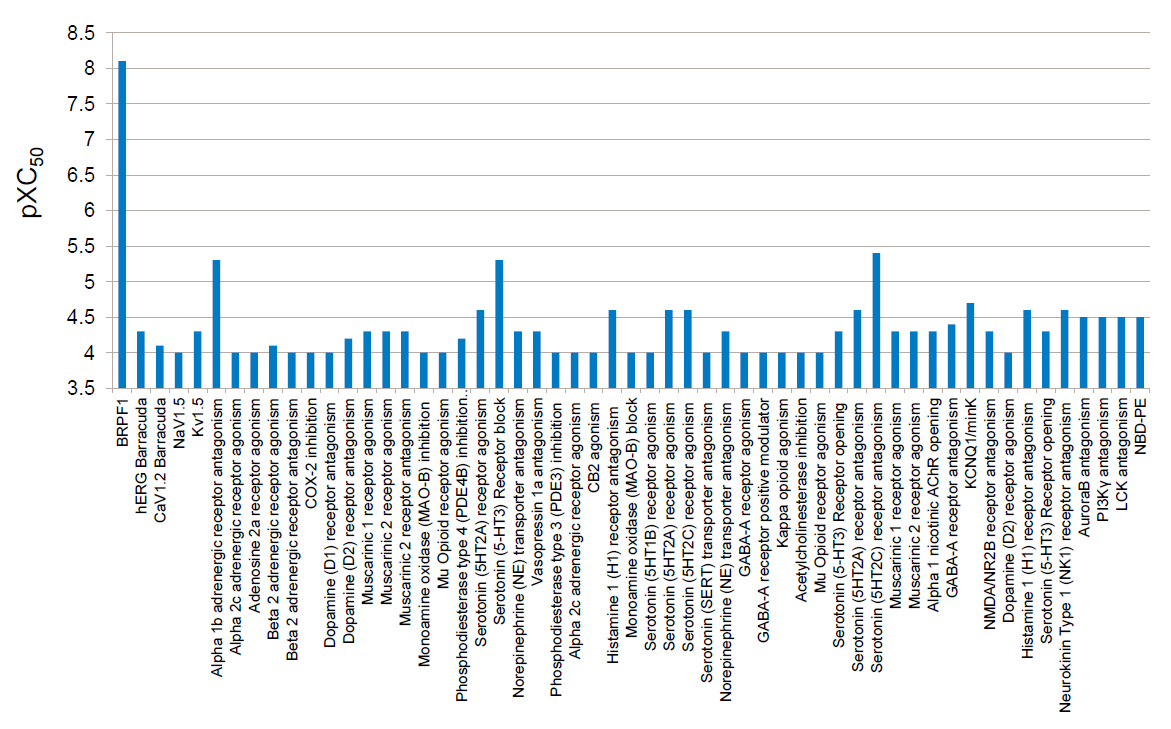
No potent off-targets identified
In a NanoBRETTM cellular target engagement assay using isolated BRPF1B BRD with NLS and Halo-tagged histone H3.3 BRPF1 showed a dose-dependent displacement from histone H3.3, with cellular IC50 of 20 nM, but no effect of the less active control GSK9311.
Chemoproteomic competition binding assay in HUT-78 cell lysate shows binding to endogenous BRPF-1 with selectivity over BRD3 (8).
Work on this probe has been published in 'GSK6853, a Chemical Probe for Inhibition of the BRPF1 Bromodomain’.
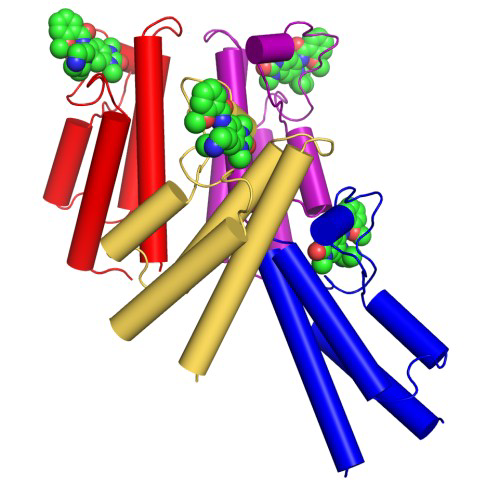
4 chains in asymmetric unit (not yet released on PDB)

X-ray structure of BRPF-1 (cyan) with GSK6853, overlayed with BRPF-2 apo (magenta) (not yet released on PDB)
NanoBRET assay
HEK293 cells (8 x 105) were plated in each well of a 6-well plate and co-transfected with Histone H3.3-HaloTag (NM_002107) and NanoLuc-BRPF1 isoform 1 (P55201-1) bromodomain amino acids 625-735 or isoform 2 (P55201-2) bromodomain amino acids 625-741. Isoform 2 has an insertion 660 -> SEVTELD in the bromodomain. Twenty hours post-transfection cells were collected, washed with PBS, and exchanged into media containing phenol red-free DMEM and 4% FBS in the absence (control sample) or the presence (experimental sample) of 100 nM NanoBRET 618 fluorescent ligand (Promega). Cell density was adjusted to 2 x 105 cells/ml and then re-plated in a 96-well assay white plate (Corning Costar #3917). Inhibitors were then added directly to media at final concentrations between 0-33 μM and the plates were incubated for 18hrs at 37 °C in the presence of 5% CO2. NanoBRET furimazine substrate (Promega) was added to both control and experimental samples at a final concentration of 10 μM. Readings were performed within 5 minutes using the CLARIOstar BMG) equipped with 450/80 nm bandpass and 610 nm longpass filters with a 0.5sec reading setting. A corrected BRET ratio was calculated and is defined as the ratio of the emission at 610 nm/450 nm for experimental samples (i.e. those treated with NanoBRET fluorescent ligand) subtracted by the emission at 610 nm/450 nm for control samples (not treated with NanoBRET fluorescent ligand). BRET ratios are expressed as milliBRET units (mBU), where 1 mBU corresponds to the corrected BRET ratio multiplied by 1000.
Chemoproteomic assay with dose-dependent competition
Chemical probe was spiked into HUT-78 nuclear and chromatin extracts and incubated for 45 min at 4 °C. Derivatized sepharose beads (35 μL beads per sample) were equilibrated in lysis buffer and incubated with cell extract pre-incubated with compound. Beads were washed with lysis buffer containing 0.2 % NP-40 and eluted with 2x SDS sample buffer supplemented with DTT. Proteins were alkylated with iodoacetamide, separated on 4–12 % NuPAGE (Invitrogen), and stained with colloidal Coomassie.