This probe is available from Cayman Chemical and Sigma.
Its negative control (FM-479) is available for purchase from Sigma.
| Probe | Negative control | |
 |
|  |
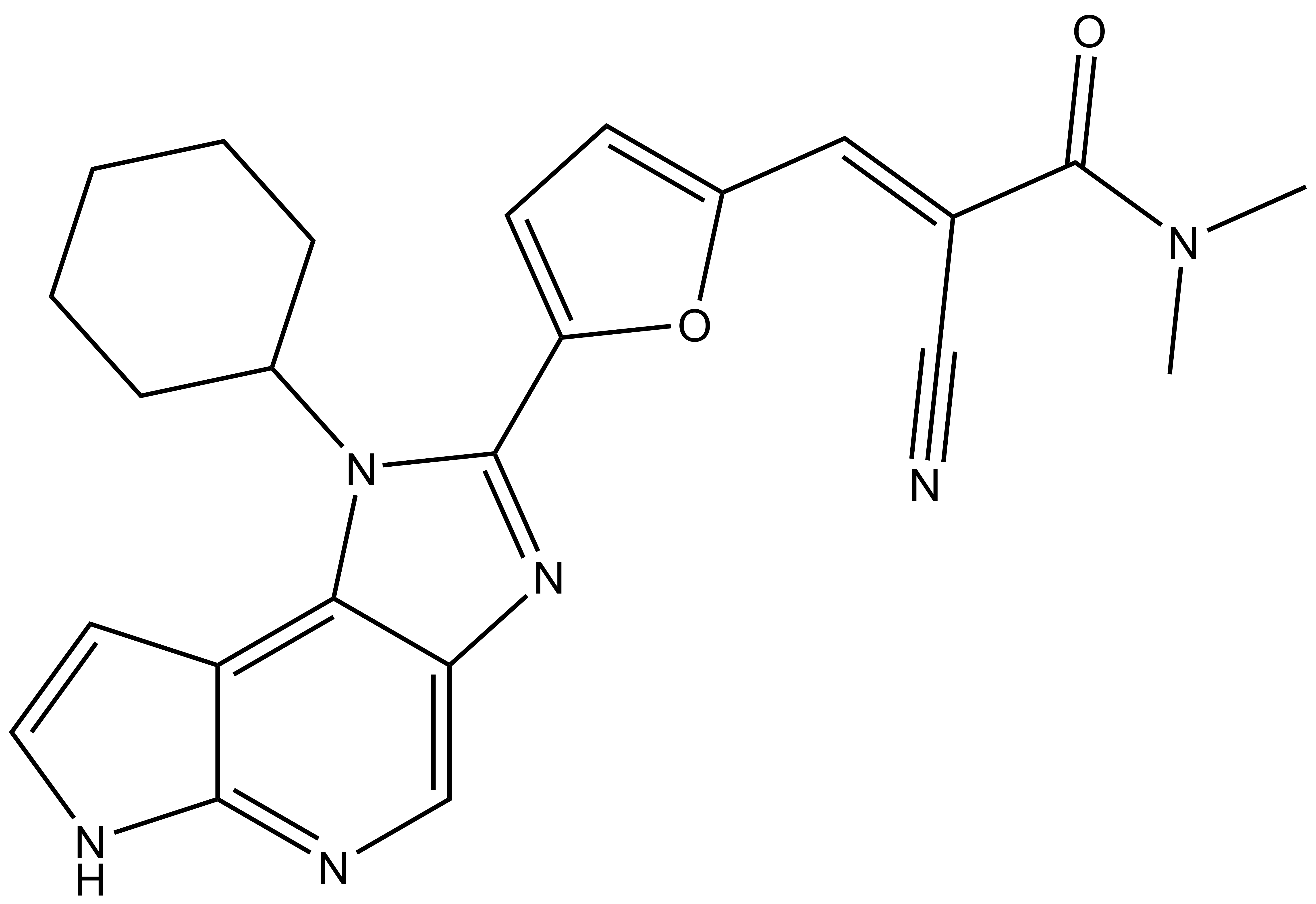
FM-381 |
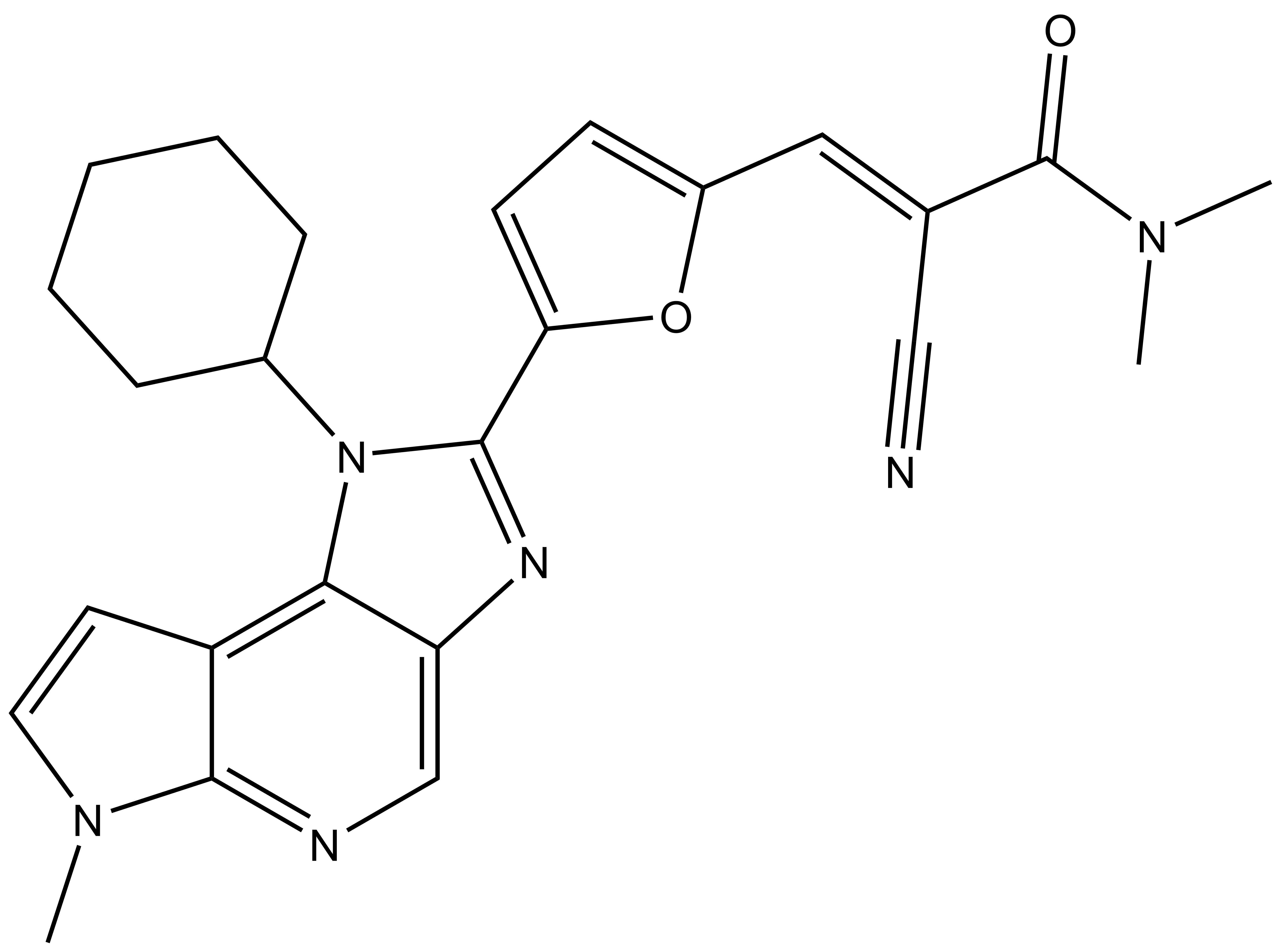
| FM-479 |
 Janus kinases (JAKs) belong to the family of cytoplasmic tyrosine kinases. In human, the JAK family itself consist of 4 members (JAK1, JAK2, JAK3 and TYK). JAK family members are multi-domain proteins of about 130 kDa, which are highly homologous with respect to their domain structure and residue conservation within their structured domains.
Janus kinases (JAKs) belong to the family of cytoplasmic tyrosine kinases. In human, the JAK family itself consist of 4 members (JAK1, JAK2, JAK3 and TYK). JAK family members are multi-domain proteins of about 130 kDa, which are highly homologous with respect to their domain structure and residue conservation within their structured domains.![]()
All JAK family members possess a catalytic kinase domain (KD) located at the C-terminus, an adjacent pseudokinase domain (PKD) flanked by a Src homology 2 (SH2) domain. The N-terminal FERM (four-point-one, ezrin, radixin and moesin homology) domain (B41) serves to mediate the interaction between the JAK and the cytokine receptor [1].
JAK kinases are effectors of the JAK-STAT signaling pathway, which is triggered by ligand binding to a cognate receptor resulting in activation of JAK by phosphorylation of key tyrosine residues within the catalytic domain. After activation, tyrosine resides in the receptor intracellular region are also phosphorylated which triggers recruitment and phosphorylation of the principal downstream effectors, the STATs. Phosphorylated STATs dimerize and translocate to the nucleus where they initiate transcription of specific responsive genes [1, 2, 3].
Contrary to the other JAK family members, JAK3 expression is restricted to the hematopoietic system, where it plays a specific role in the development of immune-competent cells [4]. This key function of JAK3 has been confirmed by loss-of-function mutations of JAK3, which cause severe combined immunodeficiency syndrome (SCID) [5]. Aberrant activation in kinase domain has been described in lymphoproliferative disorders (T-ALL; T-PLL). JAK3 is required for cytokine signalling downstream of receptors for IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21 and it acts via activation of the gamma chain receptors (γc). JAK1 and JAK3 co-localise on cytokine receptor dimers suggesting that dual JAK1 and JAK3 inhibition may be required for efficient suppression of cytokine signaling [6]. In order to study JAK3 specific functions we developed in collaboration with the laboratory of Stefan Laufer a JAK3 specific reversible covalent inhibitor.
We recommend using FM-381 at 100-300 nM for specific JAK3 inhibition. The inactive control FM-479 has no activity on JAK3 or other kinases when used at similar concentration.
As a non-covalent control inhibitor we recommend NIBR3049 (Novartis) at 1 µM (this potent reversible compound is selective for JAK3 within the JAK family, but shows also potent inhibition of other kinases such as GSK3, PKCα, PCKθ).
An additional non-covalent control compound (NIBR3049) has been donated by Novartis [7]. This inhibitor is highly selective for JAK3 within the JAK kinase family but inhibits also other kinases with considerable activity including GSK3, PKCα, PKCθ. NIBR3049 is therefore not suitable as an independent JAK3 specific probe. 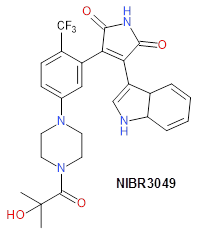
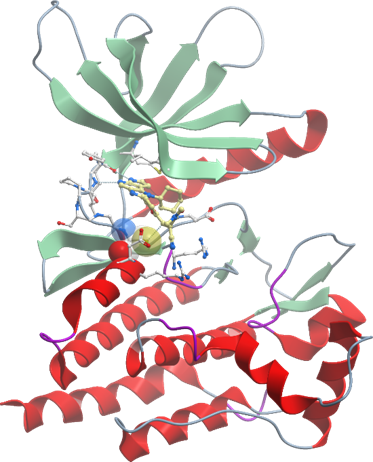
Details of the co-crystal structure of FM-381 with the kinase domain of JAK3 kinase in its reversible binding mode, click on the 'Co-Crystal structures' tab above for more details.
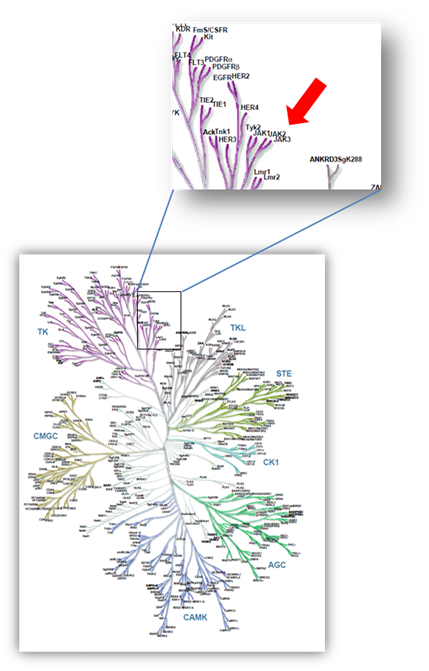
FM-381 is a potent covalent reversible inhibitor of JAK3 targeting the unique Cys909 at the gatekeeper (GK) position +7 in JAK3. FM-381 exhibited apparent JAK3 IC50 values of 0.154 nM, with 410, 2700 and 3600-fold selectivity over JAK1, JAK2 and TYK2, respectively.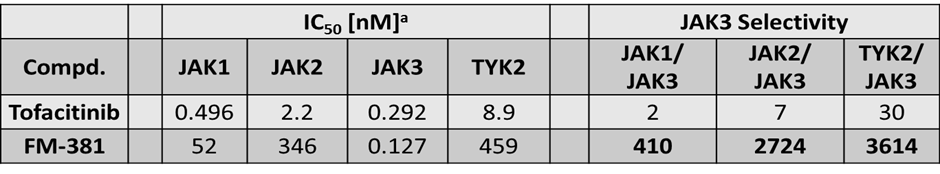
aIC50 values were calculated from the results of a radiometric assay. Data were obtained as 5-dose singlicate IC50 with 10-fold serial dilution starting at 1 µM. [ATP] = 10 µM
FM-381 shows good selectivity over other kinases with a Cys in a similar position (GK+7)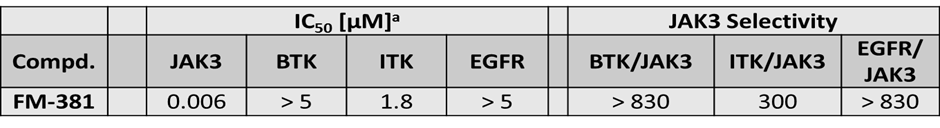
Selectivity of FM-381 was assessed in an activity assay against a panel of 410 kinases (ProQinase) at 100 nM and the compound was found to be exquisitely selective (800 fold selective over nearest kinase). Some weak interactions were observed in vitro at higher compound concentration (500 nM)
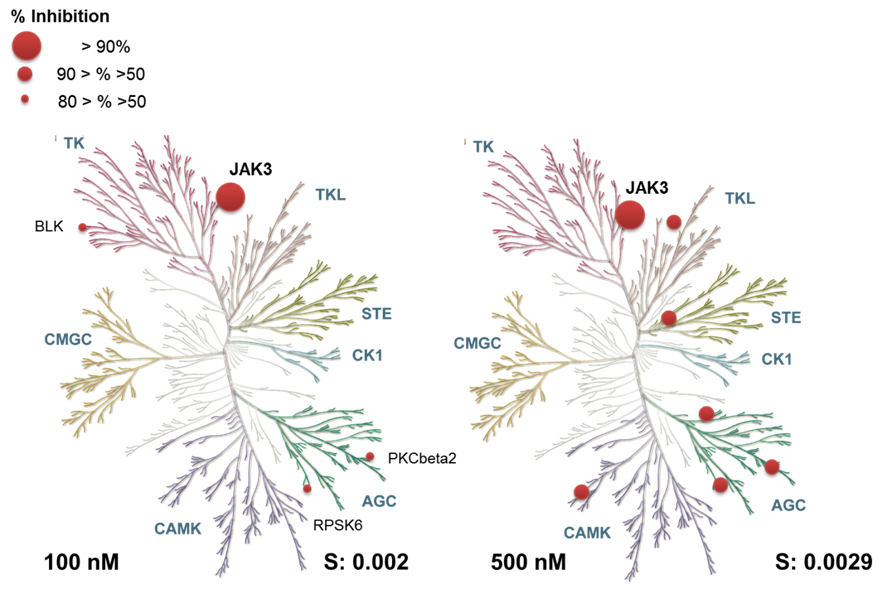
Selectivity profile of FM-381 assessed against 410 protein kinases (ProQinase). No significant activity was detected at 100 nM concentration (left panel). At 500 nM inhibitor concentration weak activity was detected (between 50 and 90% inhibition, as indicated by spheres).
FM-381 was found to be inactive in a selectivity panel of frequently hit BRDs (BRD4, BRPF, CECR, FALZ, TAF1, BRD9). Strongest hits was 500 nM for TAF1@2
FM-381 shows an apparent EC50 of 100 nM in a dose dependent BRET assay and blocks IL2 stimulated (JAK3/JAK1 dependent) STAT5 phosphorylation at 100 nM, but not JAK3 independent IL6 (JAK1/2/TYK dependent) stimulated STAT3 signalling in Human CD4+ T cells up to 1 µM.
| FM-381 |
 |
2-cyano-3-(5-(1-cyclohexyl-1,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridin-2-yl)furan-2-yl)-N,N-dimethylacrylamide Smiles: CN(C(/C(C#N)=C/C(O1)=CC=C1C2=NC3=CN=C(NC=C4)C4=C3N2C5CCCCC5)=O)C |
| Physical and chemical properties | |
|---|---|
| Molecular weight | 428.20 |
| Molecular formula | C24H24N6O2 |
| IUPAC name | 2-cyano-3-(5-(1-cyclohexyl-1,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridin-2-yl)furan-2-yl)-N,N-dimethylacrylamide |
| clogP | 4.2 |
| PSA | 71.4 |
| InChi | InChI=1S/C24H24N6O2/c1-29(2)24(31)15(13-25)12-17-8-9-20(32-17)23-28-19-14-27-22-18(10-11-26-22)21(19)30(23)16-6-4-3-5-7-16/h8-12,14,16H,3-7H2,1-2H3,(H,26,27)/b15-12+ |
| InChiKey | GJMZWYLOARVASY-NTCAYCPXSA-N |
| Storage | -20 as DMSO stock |
| Dissolution | Soluble in DMSO at least up to 50mM |
FM-381 is a potent reversible covalent inhibitor of JAK3 with an IC50 of 0.154 nM as shown in enzyme kinetic assays (ProQinase).
| IC50 [nM]a | |||||
| Compd. | JAK1 | JAK2 | JAK3 | TYK2 | |
| FM-381 | 52 | 346 | 0.127 | 459 | |
aIC50 values were calculated from the results of radiometric assay. Data were obtained as 5-dose singlicate IC50 with 10-fold serial dilution starting at 1 µM. [ATP] = 10 µM
| JAK3 Selectivity | ||
| JAK1/JAK3 | JAK2/JAK3 | TYK2/JAK3 |
| 410 | 2724 | 3614 |
Inhibition of JAK3 kinase activity was measured using a FlashPlate™-based radiometric assay (Kinase 410-Profiler, ProQinase). Data were obtained as 5-dose singlicate IC50 with 10-fold serial dilution starting at 1 µM. [ATP] = 10 µM.
ProQinase kinase assay panel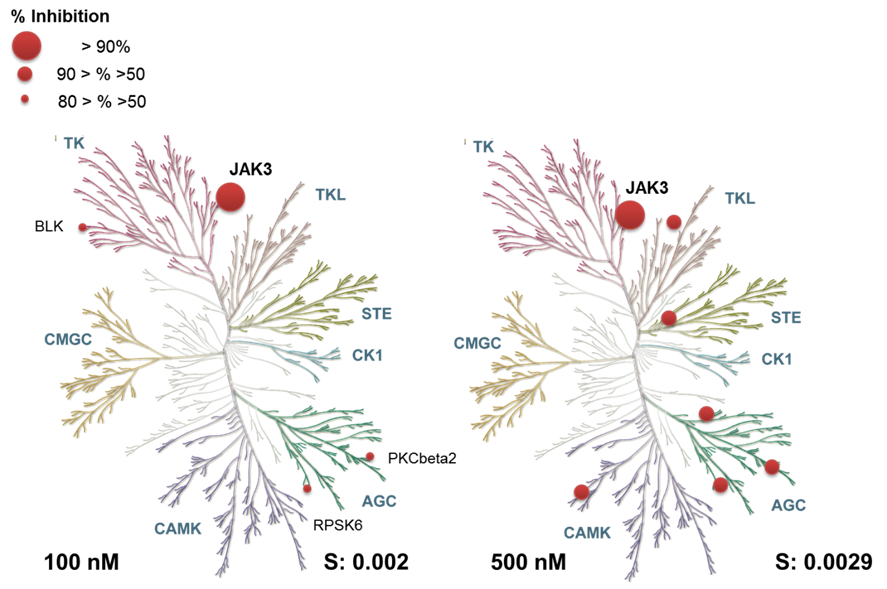
FM-381 is highly selective against 410 kinases tested at 10 µM with a selectivity score of 0.002.
| % Inhibition | ||
| 100 nM | 500 nM | |
| LRRK2 | 25 | 66 |
| MAP3K11 | 26 | 51 |
| SNARK | 28 | 61 |
| RPS6KA6 | 30 | 60 |
| PRK2 | 30 | 52 |
| BLK | 33 | 43 |
| PKC-beta2 | 39 | 69 |
| RPS6KA2 | 45 | 74 |
| JAK3 | 92 | 97 |
FM-381 shows no cross-reactivity in a selectivity panel of frequently hit BRDs by HTRF assay (BRD4, BRPF, CECR, FALZ, TAF1, BRD9). Strongest hit was 500 nM for TAF1@2.
Control compounds:
Negative control FM-479 showed no significant activity against kinome screened at 1 µM (> 8000 fold IC50). Highest activity of 82% was reported to binding to the lipid kinase PIK3C2G in DiscoverX Kinomescan). No activity was found in a selectivity panel of frequently hit BRDs by HTRF assay (BRD4, BRPF, CECR, FALZ, TAF1, BRD9) with the exception of TAF1@2 (170 nM) which results in a 1000 fold window to FM-381 on JAK3)
Positive control NIBR3049 is highly selective within the JAK/TYK family.
| Assay | NIBR3049 |
| JAK1 IC50 (nM) JAK2 IC50 (nM) JAK3 IC50 (nM) TYK2 IC50 (nM) | 1017 (> 100-fold) 2550 (> 300-fold) 8 8055 (> 1000-fold) |
NIBR3049 does show shows potent inhibition of GSK3, PKCa, PKCq, typically seen for maleimide scaffolds. Details have been reported in [7]. The compound is poorly soluble (, 0.004 g/L at pH 6.8 and only moderately cell permeable (see below).
ProQinase Selectivity panel:
FM-381 was profiled by using a Kinase 410- Profiler, a FlashPlate™-based radiometric assay (ProQinase https://www.proqinase.com). For lipid kinases and ADP-Glo™ assay technology (Promega) was used. Data were obtained as 5-dose singlicate IC50 with 10-fold serial dilution starting at 1 µM. [ATP] = 10 µM.
Binding Assay
Binding assays were performed at DiscoverX coporation in the KINOMEscan assay. Compound binding constants (Kd values) are calculated from duplicate 11-point dose-response curves using Hill equation. Curves were fitted using a non-linear least square fit with the Levenberg-Marquardt algorithm.
The cellular efficacy of FM-381 on JAK3 was assed using a Promega NanoBRET assay demonstrating on-target effect at an apparent EC50 of 100 nM in HeLa cells. The negative control compound FM-479 was negative in this assay and the positive control compound NIBR3049 showed an apparent EC50 of 1 µM. Other Cys containing kinases tested in NanoBRET assay (TEC, BTK) were negative. Only BLK showed some inhibition at micromolar concentrations. Washout experiments demonstrated that FM-381 has a residence time of ~60 min on full length JAK3 in BRET assay [4].
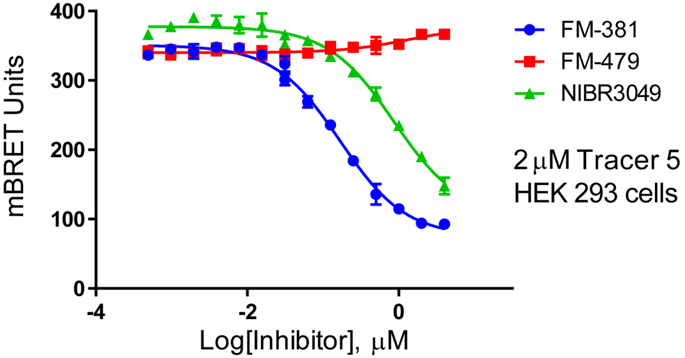
Dose dependent BRET experiment using FM-381 and the 2 control compounds showing displacement of the fluorescent tracer in C-terminally NanoLuc tagged JAK3 in HEK293 cells.
NanoBRET assay
Dose-response experiments were conducted in HeLa cells expressing NanoLuc fused to the C-terminus of full-length JAK3 using Promega tracer 5 at 2 µM in HEK293 cells in a 96 well format. A detailed description is provided in the supplemental of [4].
1. Babon JJ, Lucet IS, Murphy JM, Nicola NA, Varghese LN. The molecular regulation of Janus kinase (JAK) activation. Biochem J 2014;462(1):1-13.
2. Gurzov EN, Stanley WJ, Pappas EG, Thomas HE, Gough DJ. The JAK/STAT pathway in obesity and diabetes. FEBS J 2016;283(16):3002-3015.
3. Villarino AV, Kanno Y, Ferdinand JR, O'Shea JJ. Mechanisms of Jak/STAT signaling in immunity and disease. J Immunol 2015;194(1):21-27.
4. Forster M, Chaikuad A, Bauer SM, Holstein J, Robers MB, Corona CR, Gehringer M, Pfaffenrot E, Ghoreschi K, Knapp S et al. Selective JAK3 Inhibitors with a Covalent Reversible Binding Mode Targeting a New Induced Fit Binding Pocket. Cell Chem Biol 2016;23(11):1335-1340.
5. Pesu M, Candotti F, Husa M, Hofmann SR, Notarangelo LD, O'Shea JJ. Jak3, severe combined immunodeficiency, and a new class of immunosuppressive drugs. Immunol Rev 2005;203:127-142.
6. Haan C, Rolvering C, Raulf F, Kapp M, Druckes P, Thoma G, Behrmann I, Zerwes HG. Jak1 has a dominant role over Jak3 in signal transduction through gammac-containing cytokine receptors. Chem Biol 2011;18(3):314-323.
7. Thoma G, Nuninger F, Falchetto R, Hermes E, Tavares GA, Vangrevelinghe E, Zerwes HG. Identification of a potent Janus kinase 3 inhibitor with high selectivity within the Janus kinase family. J Med Chem 2011;54(1):284-288.
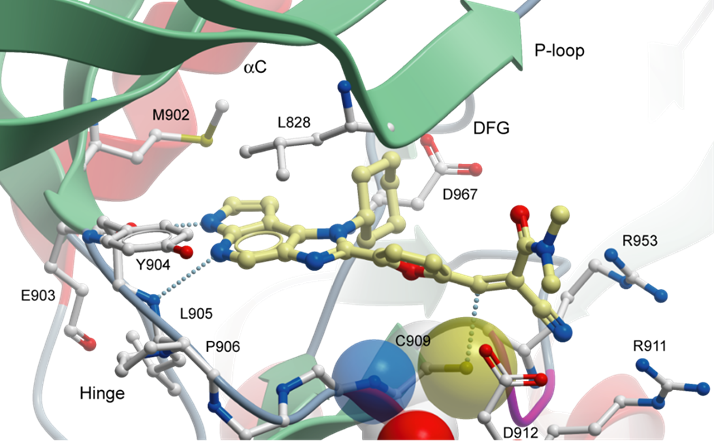
The co-crystal structures of FM-381 with the catalytic domain of JAK3 has been refined at a resolution of 1.55 Å resolution. Shown are the details of interaction of FM-381 in its non-covalent binding mode. The pyrrolopyridine hinge binding motive forms the canonical hydrogen bonds with the hinge backbone. The nitrile attached to the reactive double bond induces a pocket by re-orienting two conserved arginine residues (R953 and R911). In this structure (pdb-code: 5LWM) the covalent bond with C908 is not formed. However, the covalent as well as the non-covalent binding mode was evident in a close derivative of (FM-409; pdb-code: 5LWN). The reversible covalent interaction of FM-381 with the C908 resulted in a prolonged target residence time in cells (T1/2 ~60 min).